top of page
Search


Regulatory Horizon 2026: Navigating Innovation, Risk, and Excellence in Pharmaceuticals
Key global regulators such as the U.S. Food and Drug Administration (FDA), the European Medicines Agency (EMA), and the World Health Organization (WHO) are constantly updating their rules. This is a direct response to the increasing complexity of new therapies and global supply chains. Their goal is to ensure innovation never comes at the cost of patient safety or product quality.
Akshay Gautam
22 hours ago13 min read


Walter Healthcare Building Trust in Pharmaceutical Manufacturing Through Regulatory Excellence
This vital commitment is maintained by a strong, globally consistent system of strict monitoring, high standards, and clear accountability. This system of excellent regulation is the foundation for companies like Walter Healthcare. While some might see regulation as a complicated bureaucracy that slows things down, responsible drug manufacturers recognize it as the necessary protection. It is the crucial element that safeguards patient health and the trustworthiness of life-s
Akshay Gautam
Apr 1110 min read


Nutraceutical Playbook Part 3: India’s Nutraceutical Regulations: Safety, Science, and Market Integrity
India’s healthcare narrative is undergoing a seismic, irreversible shift. The traditional, crisis-driven model is giving way to a new focus: proactive wellness, foundational nutrition, and holistic lifestyle management. This evolution is fueled by a confluence of factors, including rising health literacy, ubiquitous digital information, the surge in lifestyle disorders, and the sharpened post-pandemic awareness that health is an asset, not an afterthought.
Akshay Gautam
Jan 3110 min read

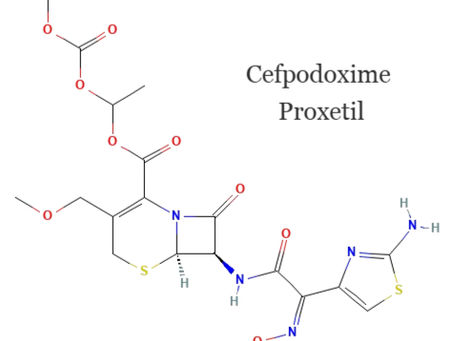
Cefpodoxime Proxetil Oral Suspension: Stability, Palatability, and Performance Redefined
This blog develops an insight about theCefpodoxime Proxetil is a prime example, standing out as a vital antibiotic in pediatrics due to its broad-spectrum efficacy, favorable safety profile, and simple dosing regimen. However, transforming the active pharmaceutical ingredient (API) into a stable, palatable, and patient-friendly oral suspension is a significant challenge in pharmaceutical engineering.
Akshay Gautam
Dec 2, 20256 min read


Faster, Stronger, Smarter Relief: Pantoprazole 40 mg + Domperidone 10 mg
This blog explores how the fixed-dose combination of Pantoprazole 40 mg + Domperidone 10 mg delivers a dual-action advantage in GERD and acid-related disorders. By combining Pantoprazole’s powerful acid suppression with Domperidone’s prokinetic action, it ensures faster relief, improved gastric motility, and long-lasting patient comfort. The article highlights its clinical superiority over monotherapy, regulatory acceptance, and formulation excellence by Walter Healthcare, ma
Akshay Gautam
Sep 13, 20255 min read


How the India–UK FTA Opens New Doors for Indian Pharma & CDMOs
The India-UK Free Trade Agreement unlocks zero-duty access and faster approvals for Indian pharma exports, paving the way for deeper...
Akshay Gautam
Aug 2, 202511 min read
Home >> Pharma News & Blog
bottom of page